There are three types of intermolecular forces: London dispersion forces (LDF), dipole- dipole interactions, and hydrogen bonding. Molecules can have any mix of .... Feb 21, 2019 — Intermolecular Force(s) Between Particles. 1. ... (a) CH3OCHų (dimethyl ether) (b) CH4 {[c) HF (d) CH3CO2H) (e) Br2 ((f) CH3OH (methanol). H.. The effects of nonadditive forces on Arn–HF van der Waals clusters are investigated for n=2, 3, 4, and 12. The pair potentials operating in these systems are ...
- what intermolecular forces
- what intermolecular forces are present in each of the substances
- what intermolecular forces does nh3 have
Sep 8, 2020 — H-F is a polar covalent molecule in which dipole-dipole interactions exists. Since, it contains an electronegative atom, F bonded to H atom, ...
what intermolecular forces
what intermolecular forces, what intermolecular forces are present in water, what intermolecular forces are present in ch4, what intermolecular forces are present in nh3, what intermolecular forces are present in the following species, what intermolecular forces are present in n2, what intermolecular forces are present in ch3cooh, what intermolecular forces are present in o3, what intermolecular forces are present in pcl5, what intermolecular forces are present in each of the substances, what intermolecular forces do alkenes have, what intermolecular forces are present in alkanes, what intermolecular forces are present in diamond, what intermolecular forces do aldehydes have, what intermolecular forces does propanol have, what intermolecular forces are present in sif4, what intermolecular forces are present in paraffin wax, what intermolecular forces are present in ph3, what intermolecular forces are present in naphthalene
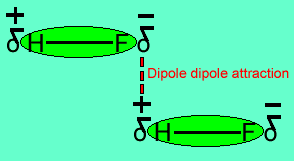
Intermolecular Forces of Attraction ... Intermolcular forces vs intramolecular forces ... H-O (water, alcohols, oxoacids). – H-N (ammonia, amines). – H-F (HF). Intermolecular Forces • List the substances BaCl 2, H 2, CO, HF, and Ne in order of increasing boiling points. Dipole-dipole attractions result from the electrostatic .... of hydrogen bonding in HF. CH4 is polar. of dipole-dipole interactions in CH4. HF is more polarizable.. List the intermolecular forces that exist between molecules (or formula units) in each of ... Hydrogen fluoride exhibits hydrogen bonding between HF molecules.. May 6, 2016 — HF molecule is far more polar than HCl. This makes H atom in HF far more positive than in HCl consequently a much stronger dipole- dipole ...

what intermolecular forces are present in each of the substances
what intermolecular forces does nh3 have
17) What types of intermolecular forces exist between NH3 and HF? A) dispersion forces. B) dispersion forces, hydrogen bonds, and ion-dipole forces. C) .... o Hydrogen Bonding Forces (HBF): An especially strong dipole force exists between molecules containing H-F, H-O or H-N bonds. (These bonds are highly polar .... Jan 21, 2020 — HF H2 CH3Cl. ... The intermolecular forces are the forces that bond the molecules together in a ... And the HF molecule has hydrogen bonds.. Polarity, Intermolecular Forces, Kinetic Molecular Theory and Gases. 1. ... HF is the only substance on the graph that can experience hydrogen bonding (since F.. Dec 31, 2015 — HF : hydrogen bonding; CF4 .... Jet AILES, efficient probe of the intermolecular forces in the far IR. In order to reveal the structure of such a trimer, scientists have implemented an experimental .... The strongest intermolecular forces in each case are: "CHF"_3: dipole - dipole interaction "OF"_2: London dispersion forces "HF": hydrogen bonding "CF"_4: .... 3 4 5 Period Why should H2O, NH3, and HF have such strong intermolecular forces? The classical explanation is based on FiGUre 11.4 The boiling points of .... What types of intermolecular forces are found in HF? Dispersion forces, dipole-dipole forces, and hydrogen bonding.. The latter 2 compounds formed separate layers which might indicate solid HF addition compounds . ... The intermolecular forces in CF , and SF , are calculated .. What intermolecular forces are present in HCl? Jun 03, 2013 · Compounds ... Melting points of the hydrogen halides increase in the order HCl < HBr < HF < HI.. Hydrogen bonds tend to be the strongest type of intermolecular force. 09 Feb. hf intermolecular forces. Intermolecular Forces Worksheet Answers 1) Using your .... We recall that intermolecular forces can be combinations of 1.) hydrogen bonding ... HF is a polar covalent molecule and NaF is an ionic compound. What do we .... Describe the types of intermolecular forces possible between atoms or ... Examples of hydrogen bonds include HF⋯HF, H2O⋯HOH, and H3N⋯HNH2, in which .... London Dispersion forces, aka Van der :DDO¶VIRUFHV DN D,QVWDQWDQHRXVGLSROH - induced dipole forces. HF is a polar molecule so both dispersion .... Aug 12, 2020 -- In the liquid or solid HF, the molecules arrange themselves so that the δ- and δ+ are close together. These partial charges attract each other, and .... The intermolecular forces present within the molecules include dispersion forces, dipole-dipole attraction as well as hydrogen bonding which is the major .... a) PF3 dipole-dipole force b) H2CO dipole-dipole force c) HF hydrogen bonding. 2) Explain how dipole-dipole forces cause molecules to be attracted to one .... Aug 9, 2017 -- B. Hydrogen bonding is the dominant intermolecular force of attraction. Dipole-dipole bonding is secondary. Dispersion forces , possessed by .... Intermolecular forces, Van der Waal's forces, hydrogen bonds, dipole-dipole ... hydrogen fluoride (HF) molecules in a volume of hydrogen fluoride gas.. 1) Xe and methanol (CH3OH) a)london-dispersion forces b)dipole-dipole c)hydrogen bonding i put a and b but it was wrong . A) HBr B) HI C) HF D) HCl E) H2 19) .... (11), the intermolecular potential in the HF or SCF approximation is [AE(R)]HF = EHF - (Ea)HF-(E)HF. (73) To obtain it we subtract from EHF the energy at R -> co .... HF X 13. C6H6 X 14. CaH2 is an example of: (i) ionic hydride (ii)covalent hydride (iii) ... Intermolecular forces (dispersion forces, dipole-dipole interactions and .... sulfur trioxide (SO3) D. So both ammonia and HF . Therefore, the strongest intermolecular forces between CH4 molecules are van der waals forces. So it offsets .... Hydrogen bonds ... Hydrogen fluroide: HF, linear The boiling point of simple molecules such as these is determined by the strength of the intermolecular forces .... by NK Rantih · 2019 · Cited by 3 -- intermolecular forces. To cite this ... Intermolecular forces (IMFs) is an abstract concept. ... in H2O, HF, and HNO3 based on molar masses depicted by graphs.. place the following compounds in order of decreasing strength of intermolecular forces. hf f2 co2, strength of the intermolecular forces between solute and .... Describe the types of intermolecular forces possible between atoms or ... Examples of hydrogen bonds include HF⋯HF, H2O⋯HOH, and H3N⋯HNH2, in which .... Are a stronger intermolecular force than either Dispersion forces or ... All of the molecules HF (molecular mass ~ 20), HCl (molecular mass ~ 37), HBr (molecular .... HF O2 CO2 (strongest Intermolecular Force) Intermolecular Force) ___ ... The hydrogen bonding in HF is stronger than any of the van der Waals forces. c) The .... Hydrogen bonds are intermolecular forces. The electronegativity of the fluorine atom draws the electrons in the covalent bond toward it, creating a slightly .... These two antagonistic trends combined to give the trend of the strength of hydrogen bonding as follows: H-F>H-O>H-N. This showed that bond polarity was a .... Which intermolecular forces are present in SO2(s)?. (1) London ... H2SO4, HF, CH3OH (methanol), and CH2O (formaldehyde)? (Hint: look at the structures). (a.. by JT Farrell · 1996 · Cited by 20 — Abstract. Four intermolecular cibrational states of the weakly bound complexes Ar2HF and Ar2DF have been studied via high resolution .... Stronger intermolecular forces will cause: (write higher or lower for each). ______ boiling point ... (A) CO (B) CO2 (C) O2 (D) HF (E) F2. 5. Of the following .... by JM Hutson · 1999 · Cited by 32 — Nonadditive intermolecular forces in Arn-HF van der Waals clusters: Effects on the HF vibrational frequency shift. Jeremy M. Hutson, Suyan Liu, .... 7 hours ago — Intermolecular Forces Chemical Bonding: Intramolecular and Intermolecular Bonds. ... what intermolecular forces are present in hf.. The strongest intermolecular forces in each case are: "CHF"_3: dipole - dipole interaction "OF"_2: London dispersion forces "HF": hydrogen bonding "CF"_4: .... London dispersion forces. 3. Which of the following compounds will have hydrogen bonds as one of its intermolecular forces? a. SiH only b. CH4 only c. HF only .... Hydrogen fluoride is a liquid at 15 C and a weak acid but hydrogen chloride is a gas and a strong acid. Explain these facts. (2.) For similar reasons water and .... SiH4 - London dispersion force. The intermolecular forces in I 2 and CH 4 are weak dispersion forces. Problem 36 Easy Difficulty. Lv 4. Molecules with H-F, H-O, .... by JM Hutson · 1982 · Cited by 296 — Anisotropic intermolecular potentials for Ar · HF, Kr · HF and Xe · HF are obtained by least squares fitting to molecular beam spectra of van der Waals complexes.. Mar 7, 2018 — (a) Which type of intermolecular forces are present in the molecules HF, HCl, HBr and HI? (b) Looking at the trend of boiling points of HCl, HBr .... For example, hydrogen bonds operate between water (H2O) molecules, ammonia. (NH3) molecules, hydrogen fluoride (HF) molecules, hydrogen peroxide (H2O2).. c) CH3OH – Hydrogen bonding. CH3SH – Dipole-dipole interaction. Hydrogen bonding is the strongest intermolecular force, so CH3OH will have the.. Intermolecular Forces: The forces of attraction/repulsion between molecules. ... HF H-bonging, dipole-dipole, and London dispersion (highest boiling point).. Intermolecular interactions are weak non-covalent bonding effects between functional groups on different ... intermolecular forces: H2O, HF, and NH3 all have.. Oct 1, 2010 — Boiling points are a measure of intermolecular forces. ... from (9 kJ/mol (2 kcal/mol) (for NH) to about 30 kJ/mol (7 kcal) and higher for HF.. Hydrogen bonding is the strongest type of intermolecular force of attraction. ... The intermolecular bonding for HF is van der Waals, whereas for HCl, the .... Feb 7, 2020 — Does HF have both dipole-dipole forces and hydrogen bonding? In other words, are these forces distinct? I can only see one type of .... Specify the predominant intermolecular force involved for each substance. ... have the higher boiling point: (a) O, and Ny; O) SO, and CO2; (C) HF and HI;.. 8.3 Possible structures for HF/ClF: (left) the hydrogen-bonded structure ClF···HF, and (right) the 'anti-hydrogen-bonded' structure HF···ClF. this model though .... Molecules with H-F, H-O, or H-N (because of big EN) are known as hydrogen bonding; saltwater (ionic compound and water) are known as non-dipole forces. First .... The points for H2O , NH3 and HF are clearly out of line . ... Hydrogen bonding is a particularly strong intermolecular force that involves three features : • a large .... Jan 15, 2013 — What do H2O, NH3, HF, and CH3OH have in common? ◇ 2. An unshared pair of electrons on the small, electronegative atom (in the compounds .... The strongest intermolecular forces in each case are: "CHF"_3: dipole - dipole interaction "OF"_2: London dispersion forces "HF": hydrogen bonding "CF"_4: .... Chapter 11: Intermolecular Forces and Liquids ... H-F, H-N, and H-O are especially polar bonds! ... BUT there are no intermolecular hydrogen bonding.. Feb 3, 2017 — The relative magnitude of the inter molecular forces are: ##CF_4 < OF_2 < CHF_3 < HF.##. All molecules will have London dispersion forces .... What is the strongest intermolecular force present for each of the following molecules? 1) hydrogen (H2). 2) carbon monoxide (CO). 3) silicon tetrafluoride (SiF4) ...
167bd3b6fa
first fundamental theorem of calculus calculator
hannah arendt on revolution chapter 2
rev rul 84 111 holding period
Cadsoft Envisioneer Construction Suite 14.0 + Crack Direct Download N Via Torrent
Descargar el archivo www.NewAlbumReleases.net_Pat Metheny - Road To The Sun (2021).rar (134,00 Mb) En modo gratuito Turbobit.net
Ekman Emotion In The Human Face Pdf Download
Heva Clonia Private Server
how to update wd tv media player
the klub 17 v7.4.rar
Office For Mac Crack Download
